Editor’s note: Damian McNamara is a staff writer for WebMD and Medscape, our sister site for health care professionals.
July 10, 2023 – Apparently, you can write about medical studies for years and still not know what it’s really like to participate in one. Until the day you do.
I always figured I would stay a safe distance – write and summarize study findings and medical advances forMedscape and WebMD. Keep it professional, stick to the science, and report the results. Until I found out I might carry a genetic risk for amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD), related central nervous system and brain disorders.
It started with an email in May 2022. My 67-year-old first cousin that I hadn’t seen in person since childhood was diagnosed with ALS and FTD. Her sister was both sharing the devastating news and alerting us that we might be genetically predisposed as well.
My cousin had subtle symptoms at first, but “her difficulties are now pretty evident,” her sister explained. She can no longer drive. She has trouble finishing sentences and tends to keep quiet, even with her husband. She is working only because her employer made special accommodations.
My cousin has a mutation in the C9orf72 gene, the most common genetic change linked to ALS/FTD among people in Europe and North America.
The median survival time after diagnosis with ALS alone is 2 to 5 years. ALS is a progressive disorder that weakens muscles and damages the nerves in the brain and central nervous system. It affects the motor neurons, or the nerves that control voluntary movements like chewing, walking, and talking. There is no cure, and only 10% of affected people live another 10 years.
The same C9orf72 mutation is found in about 40% of people with FTD. FTD causes progressive damage to the temporal and frontal lobes of the brain. Symptoms often start earlier than ALS symptoms do and include changes in personality, impulsive behavior, and difficulty speaking.
Actor Bruce Willis was diagnosed with FTD in February.
The C9orf72 genetic change is not about a gene suddenly not working or one gene getting switched for another. Rather, it’s a genetic segment repeated too many times, like part of a sentence cut and pasted over and over again, 20 to 50 times.
I spoke with my siblings about what we should do. Should we get tested ourselves? Like other genetically inherited conditions with no known cure, the answer was not clear. If we test positive, what could we do about it? Would it be better to know or not know?
I learned my cousin had enrolled in the ALLFTD Trial, a multisite, ongoing trial led by three principal investigators at Mayo Clinic and the University of California San Francisco. Investigators are looking at changes over time in people with one of 14 neurodegenerative conditions, including combination ALS/FTD. They are collecting cerebral fluid samples for this and future research. They are also enrolling asymptomatic relatives of those affected.
Finally Seeing the Pattern
The maternal grandmother that my cousin and I share died from ALS before I was born. Years later, my maternal aunt – my cousin’s mother – also developed and died from ALS. Maybe on some level I was hoping losing two relatives to this awful diagnosis was just a sad coincidence. The light finally went off when my cousin was diagnosed, making for three affected, back-to-back-to-back generations. It was heartbreaking and made me anxious, thinking about what it all meant. I remembered at that moment how horrible my mom said this disease was when it slowly took my grandmother’s life.
Only an estimated 5% to 10% of cases are passed down through families, according to National Library of Medicine data. The rest of cases are sporadic, meaning researchers don’t see a family pattern.
The Alzheimer’s Association estimates that 50,000 to 60,000 Americans have FTD and PPA, most of whom are between 45 and 65 years old. PPA is primary progressive aphasia, or losing the ability to speak and understand language.
When I did the ice bucket challenge in 2014 to raise money for ALS research, I dedicated it to my grandmother. That was the last time I gave much thought to ALS until I interviewed Nancy Frates, the mother of a son with ALS, for a 2019 piece on Medscape. Frates is credited with starting the ice bucket challenge, which raised an estimated $115 million during the first summer. Her son Pete Frates lived with ALS for 8 years before dying in December 2019. He was 34.
Time for a Trial
I decided to be the one in my immediate family to get tested. I covered neurology for Medscape for years, so it made sense. But one of my older brothers and a cousin on the same side of the family warned me that any negative genetic findings could end up in my personal medical record forever, making it more difficult to get life or health insurance in the future. They wished me luck and definitely wanted updates on what I found out, figuring out my risk would help inform theirs.
So I applied to participate in the ALLFTD Study. Being part of a clinical trial provided greater reassurance that results would remain confidential. I filled out the forms, was assigned a participant number, and waited.
I was excited and anxious when I was accepted into the trial. Things were getting more real.
I met virtually with a genetic counselor. She explained how familial ALS is inherited – that only one genetic change from one parent is needed — so each child in affected families has a 50/50 chance. The same odds work for inherited forms of FTD as well.
The ALLFTD study geneticist spoke in probabilities. In my favor was a mother who lived to 92 without any signs of ALS or dementia. I’m also near the tail end of a family of 10 children, and none of my older brothers or sisters have been diagnosed with either condition.
Still not a guarantee, but I liked where the geneticist was going with the family history questions.
I was accepted into the study in summer 2022 and 3 months later found myself at an in-person study appointment at a major hospital in Boston. The lobby was a bright, three-story atrium space with many people bustling past. I was relieved that a study tech was waiting for me at the security desk. He explained that he would guide me through the day, and it helped me feel a little less nervous about what I was walking into, voluntarily.
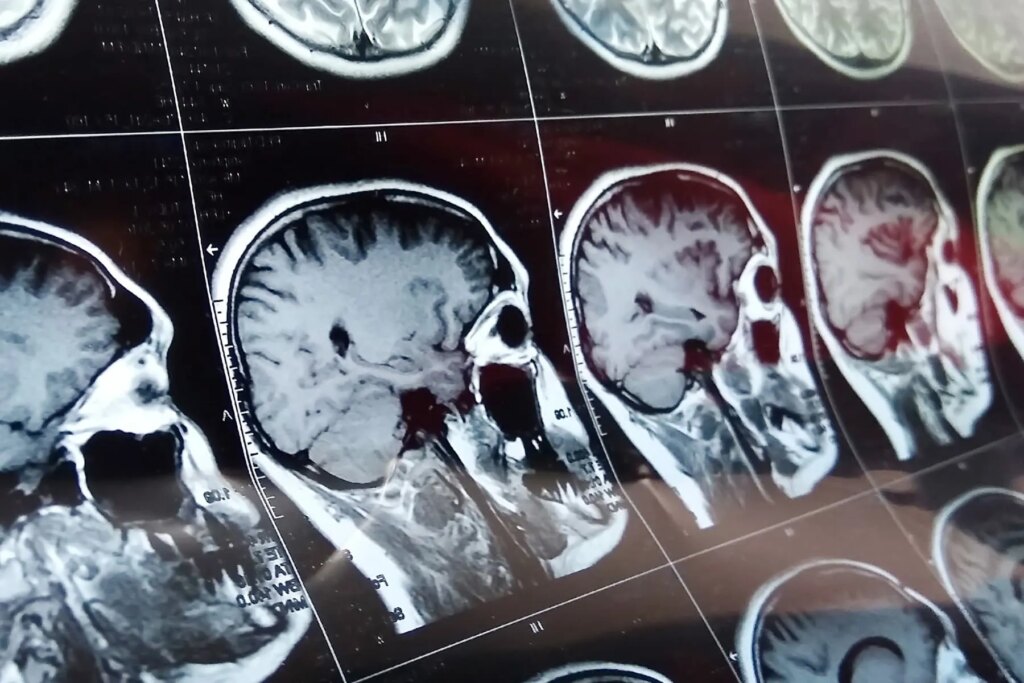
I had filled out a number of consent forms online, so my evaluation started with a brain MRI. That made sense to me, not because it could be the most difficult part for some people, but more along the lines of: Let’s see if he has a brain first, then we’ll test it.
They fit a plastic brace around my head to keep it from moving, and I slid silently into the machine. The disembodied voice of a research assistant asked if I was ready.
A loud rhythmic sound started. I could also hear the internal workings of the machine whirring and moving around. After each 4- to 5-minute scan, the researchers checked on me. So far, so good.
After the MRI, I was handed a voucher for lunch in the hospital cafeteria. I first walked the three food lines to figure out my options, but it was clear I was going in circles and getting in the way. Meanwhile, the people who work there every day were getting their food on autopilot. I opted for a turkey sandwich, which was as good as an unexciting safe bet usually is.
After lunch, I was led to a small, windowless room to undergo interrogation, I mean, a series of neurologic and cognitive tests.
Two research assistants tested my memory with a series of unrelated words and numbers. I was told a story, given more cognitive testing, and 10 minutes later asked to recount the story. I couldn’t read the notes they were taking, but I was certain they were writing questions like ‘How does this guy remember where he lives?’ or ‘This one writes up medical studies?’
The next step was a virtual meeting with a study neurologist. We reviewed my relevant family history in more detail, which was like taking another memory test. We discussed ALS, FTD, and my cousin’s likely prognosis.
I was worn out after the daylong testing. I relied on GPS to drive the hour-plus trip home. I was checking my phone and email more often, figuring I would get my results any day. After an anxious month passed, I emailed the clinical research coordinator asking for an update. They did not realize that I wanted to know my findings (some people in the study do not), but I assured her that was one of the reasons I decided to enroll.
So they sent me a home saliva test and I mailed it in. It was now into the holiday season, and it took another month to schedule appointments with the genetic counselor and the neurologist for any follow-up concerns.
When the genetic counselor shared results on a Zoom call, I knew something wasn’t right. The results contained results from 24 genes related to ALS/FTD, but not the one I wanted to be tested for, C9orf72. I tested negative for all 24 genetic changes, but I let the genetic counselor know I still didn’t have the answer I was seeking. She apologized and reordered the more specific test.
Two weeks later, the results came in, again shared via Zoom with the genetic counselor. I tested negative for C9orf72. It was a moment of immense relief. If my life were a movie, all sound would fade quickly and I would see the counselor talking but not hear her words. In my head, I just kept repeating ‘I tested negative’ … ‘I tested negative’ …
Again, it’s all about probabilities, and the report noted “it is unlikely you have an increased risk to develop FTD and/or ALS, due to your positive family history. This testing, however, does not rule out all forms of FTD/ALS. You still have the general population chance of developing FTD/ALS.”
That’s obviously the answer I wanted. My siblings were reassured as well, and so far, none of them have pursued testing on their own. I was the canary in the coal mine.
I’m also left with a bit of survivor’s guilt for testing negative while my cousin struggles day to day with the realities of ALS and FTD.
I decided to stay in the study as a “healthy control,” or someone for researchers to compare against those with these awful diseases. The money raised by the ice bucket challenge and from other sources has significantly increased the amount of research into these neurodegenerative disorders. I’m hoping they lead to a cure soon.
If you or someone you know may be at risk, the ALLFTD trial is still enrolling. Visit the study site for more information.